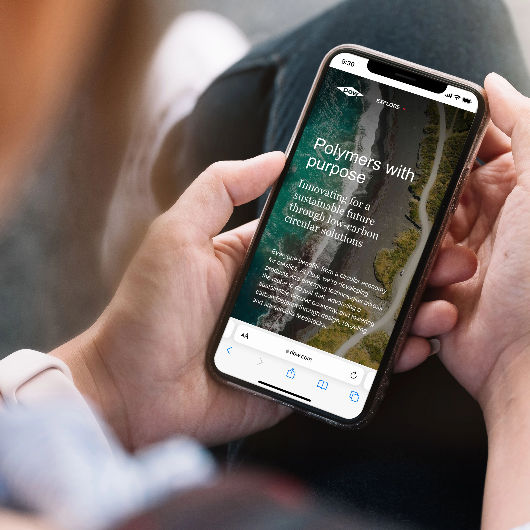
Deliver To:
- Afghanistan
- Albania
- Algeria
- Amer.Virgin Is.
- American Samoa
- Andorra
- Angola
- Anguilla
- Antarctica
- Antigua Barbuda
- Argentina
- Armenia
- Aruba
- Australia
- Austria
- Azerbaijan
- Bahamas
- Bahrain
- Bangladesh
- Barbados
- Belarus
- Belgium
- Belize
- Benin
- Bermuda
- Bhutan
- Bolivia
- Bonaire, Saba
- Bosnia-Herz.
- Botswana
- Bouvet Island
- Brazil
- Brit.Ind.Oc.Ter
- Brit.Virgin Is.
- Brunei Daruss.
- Bulgaria
- Burkina Faso
- Burundi
- C. African Rep.
- Cambodia
- Cameroon
- Canada
- Cape Verde
- Cayman Islands
- Chad
- Chile
- Christmas Islnd
- Coconut Islands
- Colombia
- Comoros
- Congo
- Cook Islands
- Costa Rica
- Cote d'Ivoire
- Croatia
- Curacao
- Cyprus
- Czech Republic
- Dem. Rep. Congo
- Denmark
- Djibouti
- Dominica
- Dominican Rep.
- Ecuador
- Egypt
- El Salvador
- Equatorial Guin
- Eritrea
- Estonia
- Ethiopia
- Falkland Islnds
- Faroe Islands
- Fiji
- Finland
- France
- Frenc.Polynesia
- French Guiana
- French S.Territ
- Gabon
- Gambia
- Georgia
- Germany
- Ghana
- Gibraltar
- Greater China (Chinese Mainland)
- Greater China (Hong Kong SAR)
- Greater China (Macau SAR)
- Greater China (Taiwan)
- Greece
- Greenland
- Grenada
- Guadeloupe
- Guam
- Guatemala
- Guinea
- Guinea-Bissau
- Guyana
- Haiti
- Heard McDon.Isl
- Honduras
- Hungary
- Iceland
- India
- Indonesia
- Iraq
- Ireland
- Israel
- Italy
- Jamaica
- Japan
- Jordan
- Kazakhstan
- Kenya
- Kiribati
- Kosovo
- Kuwait
- Kyrgyzstan
- Laos
- Latvia
- Lebanon
- Lesotho
- Liberia
- Liechtenstein
- Lithuania
- Luxembourg
- Madagascar
- Malawi
- Malaysia
- Maldives
- Mali
- Malta
- Marshall Islnds
- Martinique
- Mauritania
- Mauritius
- Mayotte
- Mexico
- Micronesia
- Minor Outl.Isl.
- Moldova
- Monaco
- Mongolia
- Montenegro
- Montserrat
- Morocco
- Mozambique
- Myanmar
- N.Mariana Islnd
- Namibia
- Nauru
- Nepal
- Netherlands
- New Caledonia
- New Zealand
- Nicaragua
- Niger
- Nigeria
- Niue
- Norfolk Island
- North Macedonia
- Norway
- Oman
- Pakistan
- Palau
- Panama
- Pap. New Guinea
- Paraguay
- Peru
- Philippines
- Pitcairn
- Poland
- Portugal
- Puerto Rico
- Qatar
- Reunion
- Romania
- Russian Fed.
- Rwanda
- S. Sandwich Ins
- S.Tome,Principe
- Saint Helena
- Saint Lucia
- Samoa
- San Marino
- Saudi Arabia
- Senegal
- Serbia
- Seychelles
- Sierra Leone
- Singapore
- Sint Maarten
- Slovakia
- Slovenia
- Solomon Islands
- Somalia
- South Africa
- South Korea
- Spain
- Sri Lanka
- St Kitts&Nevis
- St. Vincent
- St.Pier,Miquel.
- Suriname
- Svalbard
- Swaziland
- Sweden
- Switzerland
- Tajikistan
- Tanzania
- Thailand
- Timor-Leste
- Togo
- Tokelau
- Tonga
- Trinidad,Tobago
- Tunisia
- Turkey
- Turkmenistan
- Turksh Caicosin
- Tuvalu
- Uganda
- Ukraine
- United Kingdom
- United States
- Uruguay
- Utd.Arab Emir.
- Uzbekistan
- Vanuatu
- Vatican City
- Venezuela
- Vietnam
- Wallis,Futuna
- West Sahara
- Yemen
- Zambia
- Zimbabwe
Packaging
High-performance solutions

Packaging
High-performance solutions